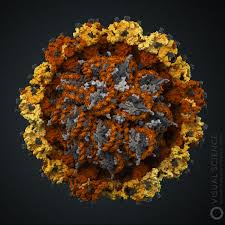
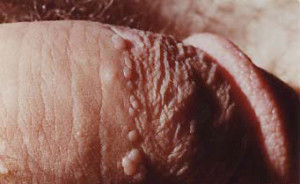
Genital warts (condylomata acuminatum) are caused by certain strains of the human papilloma virus (HPV) and resemble ordinary warts seen in other areas of the body.
HPV is the most common sexually transmitted infection throughout the world, infecting about 75% of all sexually active humans at some time in their lives. 33% of women who have just one male sexual partner will contract human papillomavirus (HPV) within a year of the relationship. The risk of infection increases with the number of partners, which varies between societies.
The presence of genital warts increases the risk of acquiring HIV by about 50%, therefore a preventative HPV vaccine will have some benefits in reducing HIV infection. (see below)
Genotypes
Of the more than 200 strains of HPV, about 40 genotypes may be passed through sexual contact. Four subtypes (16, 18, 31 and 45) have been associated with genital tract cancers, HPV-16 accounting for about 60% of cervical cancers.
Sometimes the virus is transmitted from mother to child during childbirth. (Subtypes 1, 2, 3, 4 and 10 have been shown to cause ordinary skin warts, which cannot be spread to the genitals.)
Features
Most HPV infections are transient and asymptomatic and genital warts may take 2 to 8 months to appear (sometimes many years) after the infection has been acquired. They may appear on the penis, scrotum, vulva, vaginal opening and cervix and around the anus. The time between initial HPV infection and development of cervical cancer is usually decades. Anogenital warts in children should always prompt exploration of the possibility of sexual abuse.
Obligation
Medical practitioners in Victoria have a statutory obligation under the Children, Youth and Families Act 2005 to notify the department’s Child Protection service if they believe that a child is in need of protection on the basis of sexual abuse.
HPV cannot be cultured, a Pap smear may provide the only evidence in women of infection with HPV. PCR testing from a cervical swab will determine the HPV genotype. Among newly infected women, the median time to seroconversion is approximately 8 months.
The majority of high-risk HPV infections (like other wart viruses infections) eventually disappear and do not cause cancer. The relationship between HPV16 and HPV18 and cervical cancer was demonstrated by Dr Harald zur Hausen in 1983-4.
Repeated exposure to HPVs is now recognised as the major factor in the development of cervical cancer and play an important role in cancers of the vulva, vagina, anus and oropharynx.
Vaccines, which protect against (prophylactic) and those that treat (therapeutic) human papilloma virus (HPV) are at different stages of development around the world.
Vaccine Information.
Cervical cancers represent a leading cause of deaths in women worldwide and especially in developing countries.
Every year, 300,000 women around the world die from cancer of the cervix. In 2002, 227 women died in Australia from the same disease.
HPVs are also considered a risk factor in cancer of the penis.
Currently, two prophylactic HPV vaccines Gardasil and Cervarix, utilising recombinant technology, are marketed internationally.
Vaccine anxiety
The HPV vaccine has received more parental scrutiny than most. A documentary on SBS (complete with ads) may be viewed at SBS On demand.
Vaccine efficacy
Doctors in countries using the HPV vaccine have already reported reduced numbers of young women with HPV infection or abnormal Pap smears. This reduced incidence of HPV infection in women has also benefited men, who experience reduced levels of infection through herd immunity.
PROPHYLACTIC VACCINES
The quadrivalent HPV vaccine has been classified as Category B in pregnancy.
Clinical trials of Gardasil (Merck/CSL) a recombinant vaccine offering protection against HPV types 6, 11, 16 and 18, have shown the vaccine to be 100% effective against types 16 and 18, those responsible for 70% of all cervical cancers.
Three doses are required. They may be given simultaneously (but in different arms) with hepatitis B vaccine.
The vaccine may be started from the age of 9 years, but is not recommended during pregnancy. Lactating women can receive the HPV vaccine.
Gardasil is indicated in females aged 9 to 45 years for the prevention of cervical, vulval and vaginal cancer, precancerous or dysplastic lesions, genital warts and infection caused by Human Papillomavirus (HPV) types 6, 11, 16 and 18. It is also indicated in males aged 9 to 15 years for the prevention of infection caused by Human Papillomavirus (HPV) types 6, 11, 16 and 18.
Side effects
Pain and tenderness at the site of injection. Local reactions such as redness, swelling or bruising.
Systemic side effects resemble those of placebo. Rarely, bronchospasm (difficulty in breathing) has been reported.
Precautions
Gardasil should not be given to anyone who:
– has a fever greater than 38 C.
– had a severe reaction to an earlier vaccination.
– is pregnant (defer vaccination).
Cervarix is indicated in females from 10 to 45 years of age for the prevention of cervical cancer by protecting against incident and persistent infections, cytological abnormalities including atypical squamous cells of uncertain significance (ASC-US) and cervical intraepithelial neoplasia (CIN), CIN 1 and precancerous lesions (CIN 2 and CIN 3) caused by human papillomavirus types 16 and 18. Immunogenicity studies have been conducted in females aged 10 to 14 years and 26 to 45 years to link efficacy in females aged 15 to 25 years to other populations.
Studies have confirmed the 100% efficacy and safety of this vaccine.
Cervarix is given in a 3-dose course (a 0,1 and 6 month schedule).
Side effects
Pain and tenderness at the site of injection. Local reactions such as redness, swelling or bruising.
Systemic side effects commonly include headache, myalgia, muscle weakness and fatigue. Nausea, diarrhoea, vomiting, itching, joint pain and fever are also reported.
The only serious adverse events noted with careful follow-up of over 260 000 vaccine doses has been the very rare cases of anaphylaxis (2.6 events per 100 000 doses) all of whom recovered.
Precautions
Cervarix should not be given to anyone who:
– has a fever greater than 38 C.
– had a severe reaction to an earlier vaccination.
– is pregnant (defer vaccination).

Ian Frazer
Both Gardasil and Cervarix have been been genetically engineered using viral like particles (VLPs) from HPV genotypes, by researchers, Ian Frazer and Jian Zhou, at the Centre for Immunology and Cancer Research at the University of Queensland. The recombinant L1 capsid protein from HPV self-assembles into virus-like particles, which contain no viral DNA and are therefore non-infectious. Importantly, these particles stimulate the production of antibodies that bind and neutralise the infectious virus.
The Brisbane researchers joined forces with CSL to develop a commercially viable product, used by both Merck/CSL and by GlaxoSmithKline. Vaccine details are available at the NCIRS site.
More information is available from PapScreen Victoria.
International
A five-year campaign to reach women in under-developed countries, starting with India, Peru, Uganda and Vietnam and using both Gardasil and Cervarix, has been supported by the Bill & Melinda Gates Foundation.
Merck has sort FDA approval for the expanded indication of Gardasil for the prevention of vaginal and vulval cancers.
THERAPEUTIC VACCINES
Several early trials of therapeutic vaccines have been conducted on women with existing pre-malignant changes to their cervix. So far, none have been shown to be effective in humans.
Management
The transmission of HPV during sexual intercourse is reduced by the use of condoms. Standard treatment of genital warts is aimed at destruction of the warts by physical means (freezing, diathermy, surgery), chemical means (podophyllin paint or trichloracetic acid) or through modification of the body’s immune response (Aldara cream). None are guaranteed.
BCG vaccine, commonly used to protect against TB, has been shown to clear genital warts in 80% of patients, in a recent Egyptian trial.
History of HPV vaccination in Australia.